- Link to News & Events page from breadcrumb
- Portfolio news
Portfolio news 0
Oxford Nanopore Technologies Plc
Oxford Nanopore Technologies plc - Annual results for the year ended 31 December 2025
02 Mar 2026
- Strong 2025 performance, with broad based revenue growth delivered across all regions, products and customer types and continued progress towards profitability
- Reaffirmed commitment to reach adjusted EBITDA breakeven in FY27 and become cash flow positive in FY28
Oxford Nanopore Technologies plc (LSE: ONT) (“Oxford Nanopore” or the "Group"), the company behind a new generation of molecular sensing technology based on nanopores, today announces its preliminary results for the year ended 31 December 2025.
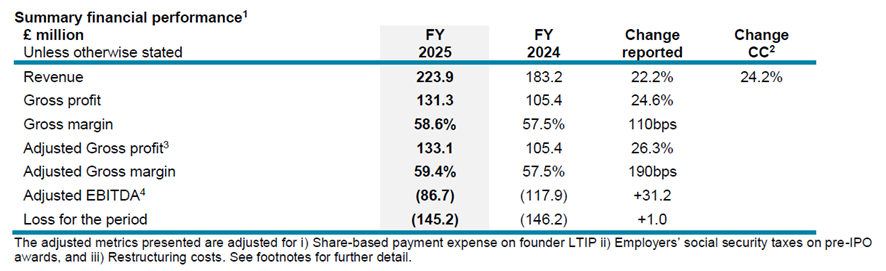
Financial highlights
- Revenue of £223.9 million grew by 24.2% on a constant currency (“CC”) basis, up 22.2% on a reported basis, slightly ahead of the top end of FY25 guidance.
- Continued commercial momentum with constant currency revenue growth above 20% in each region. EMEAI, APAC and AMR were up by 26.3% CC, 23.2% CC and 22.2% CC respectively year-on-year.
- Growth was delivered across all customer end markets; revenue grew by 59.9% in Clinical, 30.4% in BioPharma, 27.2% in Applied Industrial and 15.1% in Research.
- Revenue grew across all product categories, with fastest growth across the PromethION product range5 up 43.1% year-on-year. The MinION product range6 grew by 2.4% and Other revenue, which includes kits, services and other devices, grew by 12.0%.
- Gross margin increased by 110 basis points (bps) to 58.6% (FY24: 57.5%). Underlying margin improvements (+460bps) were driven by adoption of the new pricing model and margin improvements, particularly across PromethION Flow Cells. These gains were partially offset by a one-off non-cash inventory charge in H1 of £3.3 million (-150bps), mix (-130bps) and currency headwinds (-70bps).
- Adjusting for the impact on margin of the restructuring charge related to inventory write-downs in the year of £1.8 million (-80bps), adjusted gross margin was 59.4%.
- Adjusted EBITDA loss of £(86.7) million (FY24: £(117.9) million) with the year-on-year improvement driven by increased gross profits and ongoing disciplined control of the cost base.
- H2 adjusted EBITDA loss of £(38.4) million was £9.9 million lower than H1. This improvement in adjusted EBITDA loss is set to continue into 2026.
- Reported loss remained broadly stable at £(145.2) million (FY24: £(146.2) million), demonstrating improved operational performance and cost control, despite £22.6 million of restructuring costs incurred to realign strategic focus.
- The Group remains well capitalised with £302.8 million in cash, cash equivalents and other liquid investments7 as at 31 December 2025 (FY24: £403.8 million).
Operational and Strategic highlights
- Broad-based, growth across priority markets: Strong demand across Research and Applied markets, with particularly rapid growth in Clinical, BioPharma and Applied Industrial segments, reflecting increasing adoption where Oxford Nanopore’s richer, faster data delivers clear value over legacy and alternative approaches. Growth was delivered across all geographies, product types and customer segments, underlining the resilience and diversity of the Group’s revenue base.
- Research customers: Delivered large national and population-scale programmes. In the period, the NIHR Bioresource, Genomics England’s Cancer 2.0 and PRECISE projects scaled as expected and successfully completed, demonstrating Oxford Nanopore’s capability at delivering sequencing at scale. The UK Biobank project also transitioned from pilot to production phase to generate the first large-scale methylome dataset.
- Applied customers: Clinical growth was driven by broader adoption in infectious disease, oncology and rare disease, demonstrating a mixture of the key features and benefits of the technology, such as richer insights, turnaround time and ability to de-batch. BioPharma customers expanded use of the platform for quality control, while Industrial customers increased adoption in synthetic biology workflows.
- Progress in clinical collaborations: Entered a new strategic partnership with Cepheid, a subsidiary of Danaher, to develop and commercialise automated infectious disease sequencing solutions. Progressed collaboration with bioMérieux through the launch of AmPORE-TB, a sequencing-based solution to rapidly characterise drug-resistant tuberculosis, marking an important step in translating Oxford Nanopore technology into routine infectious disease workflows.
- Clear improvement in technology performance, workflow maturity and regulated readiness: Notable advances in throughput, cost efficiency and robustness, particularly across the PromethION platform, alongside improved basecalling, real-time methylation detection and simplified workflows, strengthened the platform’s suitability for broader adoption across research and applied settings. Milestones in regulated product development, including registration of the first IVD product, GridION Dx, and continued strengthening of quality systems and operational processes, support deeper penetration of clinical and regulated markets.
- Scientific validation underpinning commercial adoption: More than 4,000 peer-reviewed papers published in 2025 (~20,000 to date8), demonstrating the utility of Oxford Nanopore unique benefits and traction in scientific research, spanning cancer, human genetics and infectious disease, reinforcing scientific leadership and supporting continued translation into clinical and commercial settings.
- Operational discipline, scalability and redefined strategic focus: During the year, management also took steps to sharpen its strategic focus and redefine its operating model to prioritise the market opportunities and applications that best leverage Oxford Nanopore’s differentiated technology and position the business for sustainable growth. These combined actions resulted in a reduction in headcount through two restructuring events in January and November 2025, aligned with a prioritisation of R&D activities and the refinement of our product offerings to customers. These actions simplify the portfolio, concentrate investment behind the most compelling, high-priority opportunities and leave the Group better positioned for the future. In addition, the Group expanded its manufacturing and logistics capacity in 2025, introduced next-generation automated flow cell lines and continued process optimisation to enhance scalability and product stability.
- Litigation update: In the period, the Group launched patent proceedings against MGI Australia Pty Ltd. et al. (“MGI”), in which MGI has conceded that its “Cyclone SEQ WT02” infringes four of Oxford Nanopore’s Australian patents. A trial has been set for 2027 at which MGI's remaining defences will be adjudicated. Separately, in the UK Oxford Nanopore has issued proceedings in the High Court alleging trade secrets infringement, breach of confidence and breach of contract against MGI/ BGI entities. Oxford Nanopore remains confident on the value of its IP portfolio and will continue to vehemently defend its IP position when it is in the best interest of stakeholders.
- Leadership team evolution to support the next phase of growth: Today, 2 March 2026, Francis Van Parys joins the Group as CEO and Executive Director. Francis brings more than 20 years of experience leading multi-billion-dollar life science businesses, with a strong track record of scaling innovation-driven organisations through commercial and operational excellence. Previously, Francis held senior leadership roles at Radiometer (part of Danaher Corporation), Cytiva and GE Healthcare, driving sustained growth and building high-performing teams across Europe, Asia, and North America. Francis succeeds Gordon Sanghera, who has led Oxford Nanopore since its inception in 2005. Gordon will step down from the Board at close of business today, 2 March 2026, and will remain as an employee of the Group in an advisory capacity through to early 2027 to ensure a smooth handover. In addition, Tina St. Leger will join Oxford Nanopore as Chief People Officer in Q2 2026. This newly created role reflects the Group’s focus on strengthening organisational capability to support the next phase of growth, drawing on Tina’s extensive global experience across pharma and biotech, including senior leadership roles at Immunocore, GW Pharmaceuticals and GSK.
Outlook
FY26 guidance
The demand for Oxford Nanopore Technologies sensing platform remains strong and we expect to continue to outperform versus underlying end market growth in all regions.
- Revenue is expected to grow by 21-25% on a constant currency basis.
- Regionally: growth is expected to be strongest in AMR, reflecting continued progress in non-Research end-markets as we continue to navigate a stabilising, but muted, NIH market; EMEAI is expected to grow strongly, but below 2025 given the conclusion of a number of strategic projects in the Research space and timing of new projects beginning. APAC demand for ONT’s products overall remains strong but factoring in the impact from the conclusion of PRECISE II against the timing of new projects starting and ongoing specific market challenges (particularly in China) could see our growth rate moderate slightly from 2025 levels.
- The updated guidance also reflects a broader review of Oxford Nanopore’s product portfolio, including recent changes to the product range, launch timelines (particularly Q-line variants) and expected contributions.
- By end market: growth will be being weighted towards the Applied end markets (Clinical, BioPharma and Industrial).
- Gross margin is expected to be approximately 62%, driven by continued operational improvements (flow cell recycling) and tailwinds from the changes to the pricing model.
- Given the recent restructuring and continued focus on improving efficiencies in the business, overall growth in adjusted operating costs is expected to be 0-5%.
Medium term guidance update
The Group reaffirms its commitment to reach adjusted EBITDA breakeven in FY27 and become cash flow positive in FY28, reinforced by the recent restructuring events of FY25. This will be driven by:
- Above market constant currency revenue growth in FY27, which expected to be at a rate broadly similar to FY26
- Ongoing improvements to gross margins in FY27 from FY26 levels.
- Continued focus on cost discipline, with multiple levers to deliver significant operational leverage in both FY26 and FY27.
- Supported by a strong balance sheet and improving cash profile with continued focus on working capital.
Dr Gordon Sanghera, Co-founder and outgoing Chief Executive Officer, commented:
“I am pleased to report another year of strong performance for Oxford Nanopore, with revenue growth of 24.2% at constant currency, slightly ahead of the top end of our guidance, and continued progress towards profitability. Leading Oxford Nanopore for more than two decades has been an extraordinary privilege. From an idea that single-molecule sensing could be done differently, we’ve built a company that created a new category of multi-omic analysis, with a differentiated platform and expanding global customer base. I’m confident Francis will build on these strong foundations of innovation and growth to lead Oxford Nanopore into its next chapter.”
Francis Van Parys, incoming Chief Executive Officer, commented:
“I’m excited to join Oxford Nanopore today at such an important stage in its development. The company is delivering strong growth and making progress on its path to profitability, underpinned by its differentiated sensing platform and expanding global customer base. With a substantial market opportunity ahead, I look forward to building on the strong foundations established under Gordon’s leadership, enhancing commercial and operational execution, and driving innovation to deliver value for the company and for all our stakeholders.”
Presentation of results
Management will host an analyst presentation today, 2 March, at 10:45am GMT/ 5:45am EST in London.
A live webcast of the presentation will be available on Oxford Nanopore’s website at: https://nanoporetech.com/about/investors/reports.
The webcast will be recorded and a replay will be available via the same link shortly after the presentation. For further details please contact [email protected]
-ENDS-
1 Certain numerical figures included herein have been rounded. Therefore, discrepancies between totals and the sums may occur due to such rounding.
2 Constant currency (CC) applies the same rate to the FY25 and FY24 non-GBP results based on FY24 rates. All growth rates include currency fluctuations unless stated otherwise (please refer to Note 21).
3 Adjusted Gross profit is a non-IFRS measure (please refer to Note 21) that may be considered in addition to, but not as a substitute for, or superior to, information presented in accordance with IFRS. Adjusted Gross profit is the Gross profit adjusted for Restructuring costs.
4 Adjusted EBITDA is a non-IFRS measure that may be considered in addition to, but not as a substitute for, or superior to, information presented in accordance with IFRS. Adjusted EBITDA is the Loss from Operations adjusted for i) Depreciation and Amortisation ii) Share-based payment expense on founder LTIP iii) Employers’ social security taxes on pre-IPO awards, and iv) Restructuring costs. In order to reflect the core performance of the business management has redefined Adjusted EBITDA to also exclude the impacts of Other gains and losses as well as Results from associates.
5 The PromethION product range includes all PromethION devices (P2S, P2i, P24 and P48) and PromethION Flow Cells.
6 The MinION product range includes all MinION and GridION devices and MinION Flow Cells.
7 Cash, cash equivalents and other liquid investments includes cash and cash equivalents, investment bonds and UK government bonds.
8 Cumulative publications as at 31 December 2025. Note: The methodology for identifying and categorising publications has been transitioned to a new system that provides greater consistency, broader coverage and cost efficiencies, better supporting our ongoing needs. As a result of this change the prior year numbers have been restated. At 31 December 2024, cumulative publications totalled more than 16,000.
