From UK science to global scale: IP Group’s role in Pfizer’s obesity drug programme
Share:

*Published in April 2026.
The value crystallising across IP Group’s portfolio is often the result of decades of foundational science.
Idea.
Blockbuster weight loss and diabetes drugs (think Novo Nordisk’s Wegovy and Ozempic and Eli Lilly’s Zepbound and Mounjaro) have revolutionised obesity treatment by mimicking gut hormones to reduce appetite, resulting in significant weight loss and wider cardiovascular benefits. Pfizer is now preparing to enter this market with an exciting obesity drug franchise based on research carried out at UK institutions – one to which IP Group has exposure.
The scientific foundations of this story, however, stretch back more than three decades!
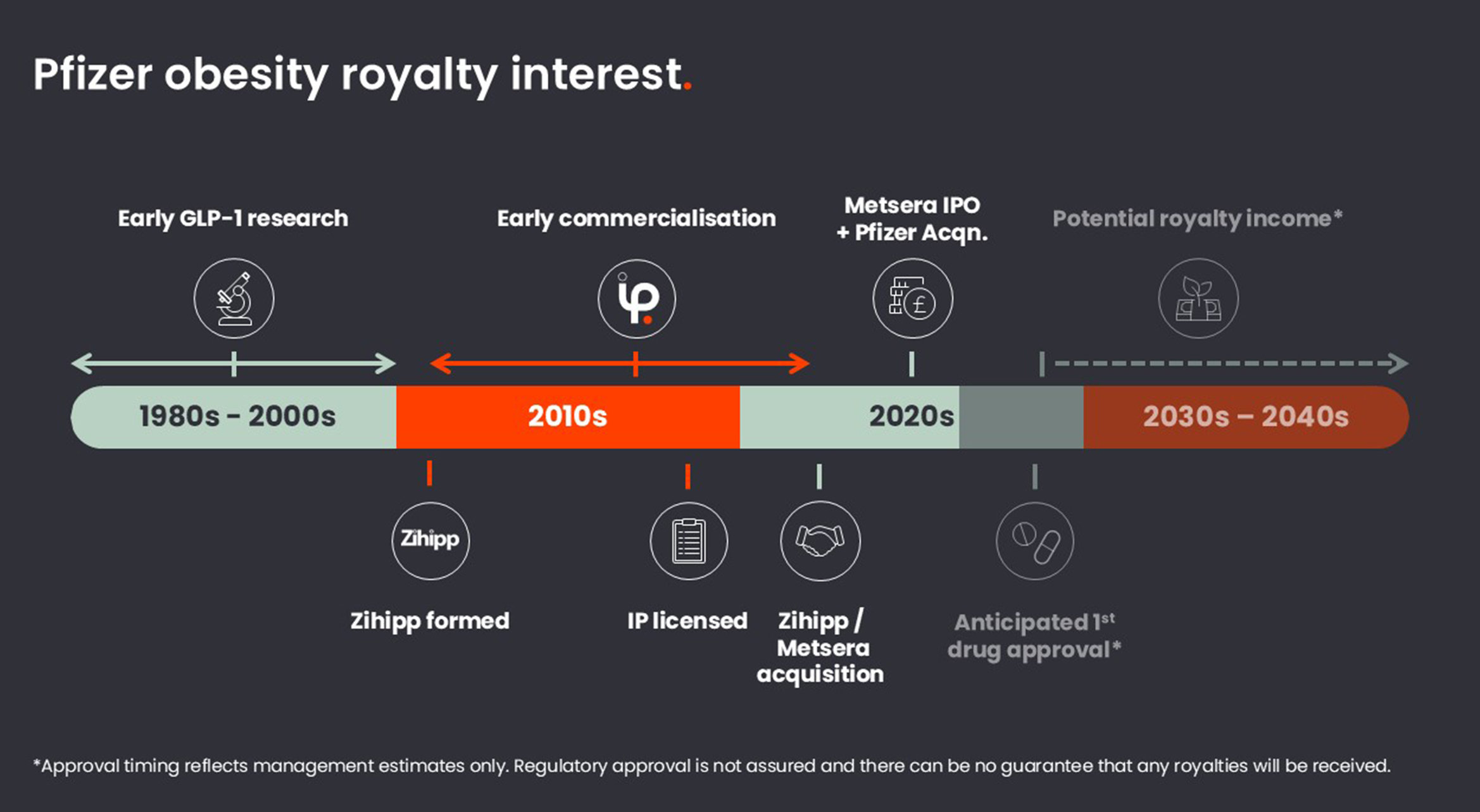
Pfizer’s lead obesity drug candidate, PF’3944 (currently in Phase III clinical trials), was invented by globally renowned Professor Sir Stephen Bloom at Imperial College London. His initial research dates back to the 1980s at the Royal Postgraduate Medical School in London, which became part of Imperial College London in 1997. In 1996, he had discovered that a hormone in the gut, known as glucagon-like peptide one (GLP-1), affects appetite, which sparked the revolution in obesity treatment using GLP-1 drugs.
As a consultant endocrinologist, I saw a lot of overweight patients who had developed diabetes. I put them on diets, which they couldn’t stick to, so I was knee-deep in the business of trying to help people eat less.
Professor Sir Steve Bloom
Nurture.
IP Group’s involvement in this story reflects a long standing partnership with Imperial College London, working closely with Professor Sir Stephen Bloom’s lab to help translate world class academic science into a commercial grade intellectual property portfolio. From the early 2010s, our focus was on helping to shape the IP strategy and ensuring the science was protected in a form capable of supporting future drug development.
A new spin-out company, Zihipp, was incorporated in 2012. Over the following years, work continued alongside Professor Sir Stephen Bloom and his team: expanding and prosecuting the patent estate, supporting early translational activity and maintaining momentum through a period when investor interest across metabolic disease remained cautious. As confidence in GLP 1–based approaches strengthened, the core IP was formally licensed into Zihipp from early 2019, providing a clear platform for future external investment, development and partnering.
IP was formally licensed into Zihipp in early 2019, providing a clear platform for future external investment, development and partnering.
That kind of early involvement is very typical of IP Group. We aim to be a long-term partner to world-class science, and we step in at points where the risk profile is often too complex for most capital.
Greg Smith, CEO, IP Group
In 2023, Zihipp was acquired by Metsera, which went on to raise c$275m through a Nasdaq listing in January 2025, valuing it at c$2bn. Following a very public and competitive bidding war, Metsera was acquired by Pfizer in 2025 for up to $10bn, giving the programme global scale and access to one of the world’s most extensive clinical, regulatory and commercial platforms.
Throughout this process and since, IP Group has acted as a long term steward of the IP, initially managing the patent portfolio and ensuring continuity as the programme scaled from UK science to global development.

Impact.
Advancing patient outcomes at global scale
More than one billion people worldwide are living with obesity and around 600 million are estimated to be living with diabetes, according to the World Health Organisation and the International Diabetes Federation. These conditions contribute to millions of premature deaths each year.
GLP 1 drugs address the underlying biology of obesity and diabetes — reducing appetite and weight, improving blood sugar control, and lowering the risk of serious complications such as cardiovascular and kidney disease.
Pfizer’s lead programme (PF’3944) is now in Phase III trials and, if successful, is targeting market approval in 2028 (subject to regulatory review), with the potential to improve outcomes for millions of patients worldwide.
Creating long‑term value for shareholders
For IP Group shareholders, this journey has created exposure to a long‑dated royalty interest being developed by one of the world’s best‑resourced pharmaceutical companies and linked to a major global healthcare market.
First, we now have exposure to a long-dated royalty interest that’s being developed by one of the world’s best-resourced pharma companies and is linked to one of the most significant pharma markets of the coming decades…Secondly, we have recognised a fair value of £128m for that interest at the 2025 year-end.
Greg Smith, CEO, IP Group
IP Group benefits from financial exposure to a number of the Pfizer obesity drug programmes and is entitled to receive future returns through a combination of technical and commercial milestone payments, as well as tiered, low-single digit percentage royalties on net sales of licensed products. 50% of all monies received by the Group are payable to Imperial College London under revenue share arrangements, with any future royalties subject to the approval and launch of the relevant drugs.
IP Group recognised a fair value of £128m for its interest (net of the share payable to Imperial College London) in its 2025 results, reflecting the discounted value of expected future milestone and royalty income. Should the drugs to which the Group has exposure to successfully progress through trials to product launch, the Group expects this value could increase significantly. IP Group’s financial exposure comes from both the IP licence and the Group’s previous shareholding in Zihipp.
With no future development costs to bear, the asset offers an asymmetric risk‑reward profile — demonstrating how early, patient engagement with world‑class science can generate enduring value for both society and shareholders.
Forward looking statements: This web page may contain forward looking statements based on current expectations and assumptions, which involve risks and uncertainties. Actual results may differ materially. Past performance is not a guide to future performance. Forward looking statements speak only as at the date made, and the Company undertakes no obligation to update them, except as required by law. Nothing on this webpage constitutes a profit forecast.
